Our Research
BioMEL aims to advance the fields of regenerative engineering and immunoengineering by focusing on engineering the physicochemical properties of biomaterials to allow them to directly modulate cell and tissue responses. These novel biomaterials, defined as biomodulatory materials, are being utilized individually or in combination with bioactive factors to produce desirable biomedical outcomes. Specifically, biomodulatory materials are being created to regenerate complex tissues through improved vascularization and innervation, exploit the immune system to enhance regenerative engineering, and induce immunological responses through targeted peptide delivery.
Immune-Mediated Regeneration
A significant barrier for biomaterials-based regenerative engineering is the host immune response. Even benign constructs have been shown to induce significant inflammation and recruit immune cells. A common approach to solving this problem has been the use of stealth elements to keep the immune system from recognizing the biomaterial. While somewhat promising results have been seen, this technique avoids the fundamental link that exists between activation of the immune response and tissue remodeling. When a tissue is damaged naturally, the immune system works to debride the area and stimulate the body’s natural regenerative machinery. By better understanding and exploiting the host immune response to biomaterials, it is possible to develop novel and innovative immunoregenerative engineering solutions.
The goal of this research is to characterize and modulate the immune response to biomodulatory materials-based systems. It is hypothesized that through physicochemical materials modulation and bioactive factor incorporation, complex constructs can be generated that better facilitate tissue regeneration.
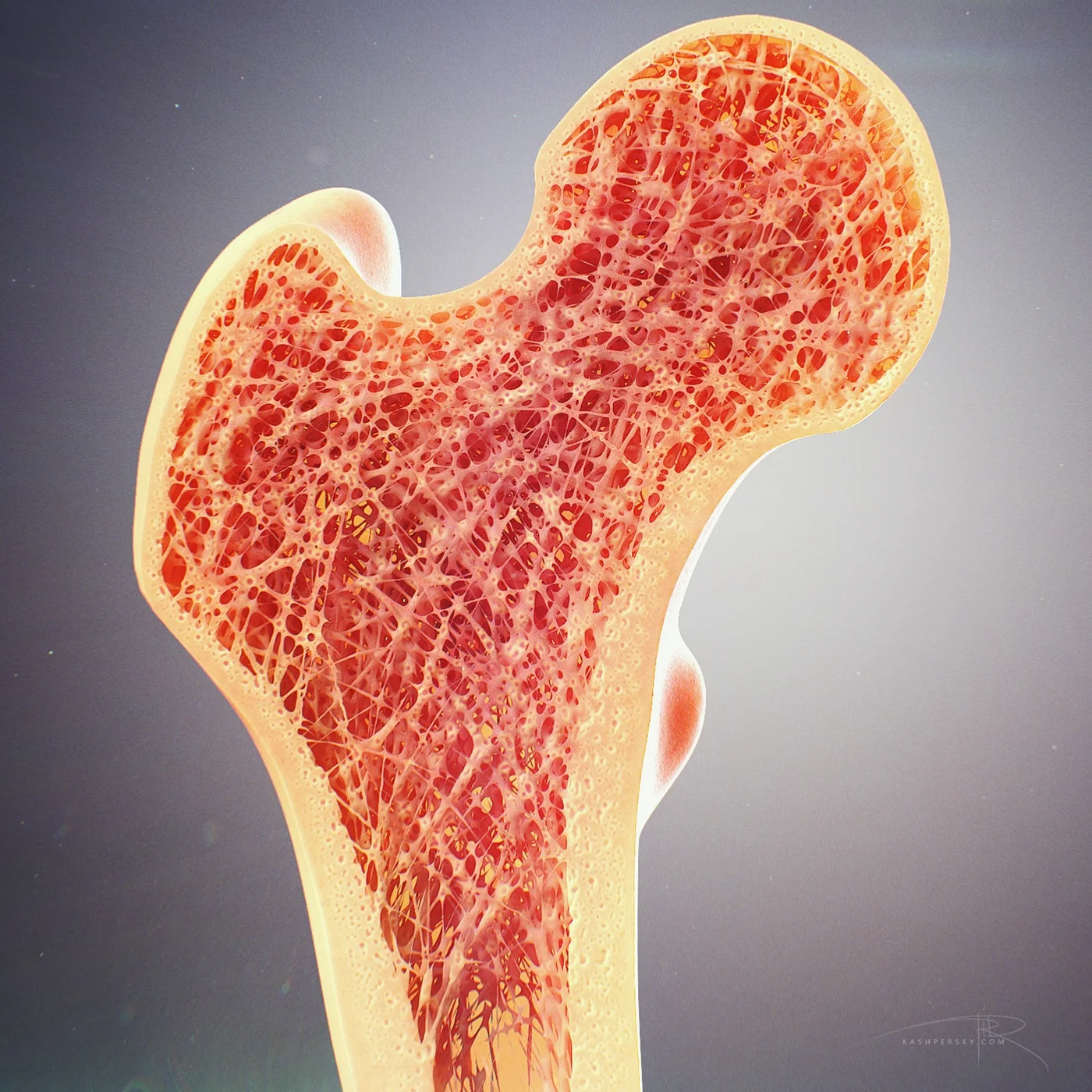
Complex Tissue Regeneration
Improvements in construct vascularization and innervation can benefit several regenerative engineering subfields, with musculoskeletal regeneration being of particular importance. Many musculoskeletal tissues are vascularized to facilitate nutrient delivery, waste removal, and cell turnover; and innervated to allow for development and remodeling. Often when regenerative engineering constructs are designed, significant attention is paid to the musculoskeletal cells of interest, but little thought is given to replicating the vascular and nerve networks of the host tissue. This has led to many studies in which scaffolds are well tolerated in vivo, but only facilitate host tissue regeneration at the periphery of the implant. Recent studies have shown a direct correlation between musculoskeletal, vascular, and nerve tissue induction that can be exploited to improve regenerative engineering strategies.
The goal of this research is to design complex biomodulatory materials-based systems capable of facilitating vascularized and innervated tissue regeneration. It is hypothesized that this can be achieved by tuning the physicochemical properties of materials to create novel, multifaceted scaffolds.
Targeted Peptide Delivery
Communicable diseases and cancer are the second and third leading causes of death in the world, respectively. The most common strategy utilized to manage these diseases is the employment of small molecule therapies. While these have been quite successful, they have led to the rise of treatment-resistant organisms and cancer cells. A possible alternative method is to treat with a more specific and harder to resist therapeutic modality and/or prevent the disease through vaccination. Both of these strategies commonly rely on the use of peptides which possess significant advantages over small molecule therapeutics, especially in their specificity. By developing biomodulatory materials-based delivery systems, a new platform technology for preventing and/or treating diseases can be realized.
The goal of this research is to create biomodulatory materials capable of prophylactically or therapeutically targeting the immune response to prevent or treat infectious diseases and cancer. It is hypothesized that this can be achieved by creating peptide amphiphile micelles with targeting modalities to facilitate host peptide delivery to specific cells and tissues of interest.